 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Belsomra |
| Other names | MK-4305 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a614046 |
| License data |
|
| Routes of administration | By mouth |
| Drug class | Dual orexin receptor antagonist (DORA)[1] |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 82% (at 10 mg) |
| Protein binding | >99% |
| Metabolism | hepatic, CYP3A, CYP2C19 |
| Elimination half-life | ~12 hours |
| Excretion | Feces (66%), urine (23%) |
| Identifiers | |
| |
| Chemical and physical data | |
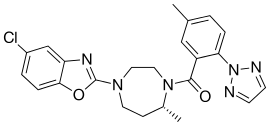
| Formula | C23H23ClN6O2 |
| Molar mass | 450.93 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Suvorexant, sold under the trade name Belsomra, is a medication used to treat trouble sleeping, including trouble falling asleep and staying asleep.[1] It may be effective for at least a year.[1] It is taken by mouth, half an hour before bed.[1]
Common side effects include sleepiness, headache, unusual dreams, cough, and dry mouth.[1] Other side effects include abuse, sleep paralysis, suicide, anxiety, and a decreased ability to drive.[1] Safety in pregnancy and breastfeeding is unclear.[3] It is a dual orexin receptor antagonist (DORA).[1]
Suvorexant was approved for medical use in the United States in 2014,[1] and Canada in 2018.[4] In the United States it costs about 380 USD per month as of 2021.[5] In the United States it is a Schedule IV controlled substance.[6]
References edit
- ^ a b c d e f g h i j k "Suvorexant Monograph for Professionals". Drugs.com. Archived from the original on 7 March 2016. Retrieved 15 October 2021.
- ^ "Suvorexant (Belsomra) Use During Pregnancy". Drugs.com. 9 September 2019. Archived from the original on 24 January 2021. Retrieved 30 January 2020.
- ^ "Suvorexant (Belsomra) Use During Pregnancy". Drugs.com. Archived from the original on 24 January 2021. Retrieved 15 October 2021.
- ^ "Regulatory Decision Summary - Belsomra - Health Canada". hpr-rps.hres.ca. Government of Canada. Archived from the original on 6 July 2020. Retrieved 6 February 2020.
- ^ "Suvorexant Prices, Coupons & Savings Tips - GoodRx". GoodRx. Retrieved 15 October 2021.
- ^ "Rules - 2014 - Final Rule: Placement of Suvorexant into Schedule IV". www.deadiversion.usdoj.gov. Archived from the original on 2016-04-17. Retrieved 2016-04-03.