 | |
| Clinical data | |
|---|---|
| Trade names | Wakix, Ozawade, others |
| Other names | Tiprolisant; Ciproxidine; BF2.649 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a619055 |
| License data |
|
| Routes of administration | By mouth |
| Drug class | Histamine 3 (H3) receptor inhibitor[1] |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| Chemical and physical data | |
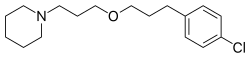
| Formula | C17H26ClNO |
| Molar mass | 295.85 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Pitolisant, sold under the brand name Wakix among others, is a medication for the treatment of excessive daytime sleepiness in narcolepsy.[1] It is taken by mouth once a day in the morning.[3][4]
Common side effects include headache, trouble sleeping, nausea, and anxiety.[3] Other side effects may include QT prolongation.[5] Safety in pregnancy is unclear.[6] It bring about an opposite to usual response at the histamine 3 (H3) receptor.[1] This increases the activity of histamine neurons in the brain, which keeps a person awake.[7][3]
Pitolisant was approved for medical use in the United States in 2019 and Europe in 2021.[5][3] In the United Kingdom it costs the NHS about £310 per month as of 2021.[4] In the United States this amount costs about 6,800 USD.[8]
References edit
- ^ a b c d e "Wakix- pitolisant hydrochloride tablet, film coated". DailyMed. 6 November 2019. Archived from the original on 11 August 2020. Retrieved 18 August 2020.
- ^ "Wakix EPAR". European Medicines Agency (EMA). Archived from the original on 12 November 2020. Retrieved 18 August 2020. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ a b c d e f "Ozawade EPAR". European Medicines Agency (EMA). Archived from the original on 15 October 2021. Retrieved 15 October 2021.
- ^ a b c BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 513. ISBN 978-0857114105.
- ^ a b "Pitolisant Monograph for Professionals". Drugs.com. Archived from the original on 21 January 2021. Retrieved 28 October 2021.
- ^ "Pitolisant (Wakix) Use During Pregnancy". Drugs.com. Archived from the original on 18 March 2020. Retrieved 28 October 2021.
- ^ Syed YY (20 July 2016). "Pitolisant: First Global Approval". Drugs. 76 (13): 1313–1318. doi:10.1007/s40265-016-0620-1. PMID 27438291. S2CID 42684839.
- ^ "Wakix Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 24 January 2021. Retrieved 28 October 2021.