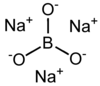
Trisodium borate is a chemical compound of sodium, boron, and oxygen, with formula Na3BO3, or (Na+)3[BO3]3−.[3] It is a sodium salt of the orthoboric acid B(OH)3.
| |
| Names | |
|---|---|
| IUPAC name
Trisodium orthoborate
| |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| 117865 | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Na3BO3 | |
| Molar mass | 127.78 g·mol−1 |
| Density | 1.73 g/cm3[1] |
| Melting point | 75 °C (167 °F; 348 K) [1] |
| Boiling point | 320 °C (608 °F; 593 K) [1] |
| Hazards | |
| GHS labelling:[2] | |
 
| |
| Warning | |
| H319, H360, H361d, H412 | |
| P203, P264+P265, P273, P280, P305+P351+P338, P318, P337+P317, P405, P501 | |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
The compound is also called trisodium orthoborate, sodium orthoborate, or just sodium borate. However, "sodium orthoborate" has been used also for a compound with formula Na4B2O5, which would correspond to an equimolar mixture of sodium metaborate NaBO2 and trisodium borate proper.[4] and "sodium borate" is sometimes used in the generic sense, for a sodium salt with any of several other borate anions.
Preparation
editSodium carbonate Na2CO3 will react with sodium metaborate NaBO2 or boric oxide B2O3 to form the orthoborate and carbon dioxide when heated between 600 and 850 °C:[3]
- NaBO2 + Na2CO3 → Na3BO3 + CO2
Difficult to obtain in pure form from melts.[5]
Properties
editThis section is empty. You can help by adding to it. (August 2022) |
Reactions
editWhen dissolved in water, the orthoborate anion partially hydrolyzes into metaborate [BO2]− and hydroxide OH−:[3]
- [BO3]3− + H2O ⇌ [BO2]− + 3 OH−
Electrolysis of a solution of sodium orthoborate generates sodium perborate at the anode.[6] [7]
References
edit- ^ a b c "Trisodium borate" Product page in the World Of Chemicals website. Accessed on 2022-06-27.
- ^ "Boric acid, sodium salt". pubchem.ncbi.nlm.nih.gov.
- ^ a b c Pasupathy Rajan Subbaiyan (2003): "Study of Trisodium Borate Formation and Its Reaction with Green Liquor in Partial Autocausticizing". Masters Thesis, Western Michigan University.
- ^ Daniel L. Calabretta and Boyd R. Davis (2007) "Investigation of the anhydrous molten Na–B–O–H system and the concept: Electrolytic hydriding of sodium boron oxide species". Journal of Power Sources, volume 164, issue 2, pages 782-791. doi:10.1016/j.jpowsour.2006.11.023
- ^ G. W. Morey and H. E. Merwin (1936): "Phase Equilibrium Relationships in the Binary System, Sodium Oxide-Boric Oxide, with Some Measurements of the Optical Properties of the Glasses". Journal of the American Chemical Society, volume 58, issue 11, pages 2248–2254. doi:10.1021/ja01302a04
- ^ Wilfrid Gustav Polack (1915): "The anodic behaviour of alkaline borate and perborate solutions". Transactions of the Faraday Society, volume 10, pages 177-196. doi:10.1039/TF9151000177
- ^ Tanatar (1898): Zh. Pys. Chem., volume 26, page 132.