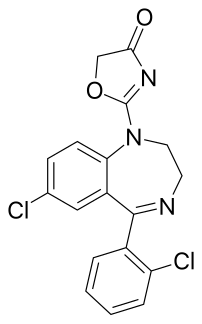
Reclazepam is a drug which is a benzodiazepine derivative. It has sedative and anxiolytic effects similar to those produced by other benzodiazepine derivatives, and has a short duration of action.[1]
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C18H13Cl2N3O2 |
| Molar mass | 374.22 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Synthesis
editThe reduction of the lactam in Delorazepam with lithium aluminium hydride gives CID:20333776 (1). Condensation with 2-chloroacetylisocyanate [4461-30-7] (2) proceeds to afford urea, CID:20333773 (3). Reaction of that with sodium iodide and base probably proceeds initially by halogen exchange of iodine for chlorine (Finkelstein reaction). Subsequent replacement of iodide by the enol anion of the urea oxygen results in formation of the oxazolone ring. There is thus obtained reclazepam (4).
See also
editReferences
edit- ^ "Reclazepam". Pscyhotropics.dk. 20 January 1986. Retrieved 24 August 2009.[dead link]
- ^ US 4208327, Yonan PK, "5-Aryl-1-(2-oxazolin-2-yl)-1H-1,4-benzodiazepines and related compounds", issued 17 June 1980, assigned to GD Searle LLC.
- ^ Speziale AJ, Smith LR (April 2003). "α‐Chloroacetyl Isocyanate: Isocyanic acid, anhydride with chloroacetic acid". Organic Syntheses. 46: 16. doi:10.1002/0471264180.os046.05.
- ^ US 3144439, Reeder E, Sternbach LH, "Process for production of benzodiazepines", issued 1964, assigned to Hoffmann-La Roche Inc