Prednicarbate is a relatively new topical corticosteroid drug. It is similar in potency to hydrocortisone. Corticosteroids have always been an important part of the pharmacological arsenal of dermatology; however, their tendency to produce side-effects has caused the need to search for new preparations.[1]
 | |
| Clinical data | |
|---|---|
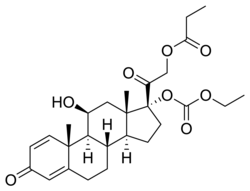
| Other names | {2-[(8S,9S,10R,11S,13S,14S,17R)-17-ethoxycarbonyloxy-11-hydroxy-10,13-dimethyl-3-oxo-7,8,9,11,12,14,15,16-octahydro-6H-cyclopenta[a]phenanthren-17-yl]-2-oxoethyl} propanoate |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a604021 |
| Routes of administration | Topical |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.070.516 |
| Chemical and physical data | |
| Formula | C27H36O8 |
| Molar mass | 488.577 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
It is nonhalogenated.[2]
References
edit- ^ United States Pharmacopeia, the National Formulary. United States Pharmacopeial Convention, Incorporated. 2008. p. 3060. ISBN 978-1-889788-53-1.
- ^ Gupta AK, Chow M (2004). "A review of prednicarbate (Dermatop)". Skin Therapy Lett. 9 (10): 5–6, 9. PMID 15657633.