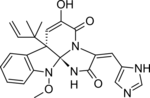
Meleagrin and its derivatives such as oxaline are bio-active benzylisoquinoline alkaloids made by various species of Penicillium fungi.[1] It is similar to other fungal alkaloids, such as Roquefortine C, which is made as an intermediate in the same biosynthetic pathway.
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C23H23N5O4 | |
| Molar mass | 433.468 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
It was suggested to have inhibitory activity on fatty acid synthesis for the bacteria Staphylococcus aureus and Streptococcus pneumoniae.[2]
Biosynthesis edit
The biosynthetic pathway was determined to contain several genes, including a non-ribosomal peptide synthetase.[3] The biosynthesis begins by cyclizing the two amino acids histidine and tryptophan, and is the followed by the addition of an isoprene, and several ring rearrangement steps.[4]
References edit
- ^ Du, L; Feng, T; Zhao, B; Li, D; Cai, S; Zhu, T; Wang, F; Xiao, X; Gu, Q (April 2010). "Alkaloids from a deep ocean sediment-derived fungus Penicillium sp. and their antitumor activities". The Journal of Antibiotics. 63 (4): 165–70. doi:10.1038/ja.2010.11. PMID 20186171. S2CID 12744541.
- ^ Zheng, CJ; Sohn, MJ; Lee, S; Kim, WG (2013). "Meleagrin, a new FabI inhibitor from Penicillium chryosogenum with at least one additional mode of action". PLOS ONE. 8 (11): e78922. Bibcode:2013PLoSO...878922Z. doi:10.1371/journal.pone.0078922. PMC 3842914. PMID 24312171.
- ^ Ali, Hazrat; Ries, Marco I.; Nijland, Jeroen G.; Lankhorst, Peter P.; Hankemeier, Thomas; Bovenberg, Roel A. L.; Vreeken, Rob J.; Driessen, Arnold J. M. (12 June 2013). "A Branched Biosynthetic Pathway Is Involved in Production of Roquefortine and Related Compounds in Penicillium chrysogenum". PLOS ONE. 8 (6): e65328. Bibcode:2013PLoSO...865328A. doi:10.1371/journal.pone.0065328. PMC 3680398. PMID 23776469.
- ^ García-Estrada, Carlos; Ullán, Ricardo V.; Albillos, Silvia M.; Fernández-Bodega, María Ángeles; Durek, Pawel; von Döhren, Hans; Martín, Juan F. (23 November 2011). "A Single Cluster of Coregulated Genes Encodes the Biosynthesis of the Mycotoxins Roquefortine C and Meleagrin in Penicillium chrysogenum". Chemistry & Biology. 18 (11): 1499–1512. doi:10.1016/j.chembiol.2011.08.012. PMID 22118684.