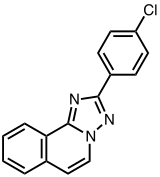
Lotrifen (INN), also known as trazisoquine and sold under the brand names Canocenta and Privaprol, is an abortifacient which is used in veterinary medicine.[1][2] It was described in 1978.[1]
 | |
| Clinical data | |
|---|---|
| Trade names | Canocenta, Privaprol |
| Other names | Trazisoquine; L-12717; DL-717-IT |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.060.340 |
| Chemical and physical data | |
| Formula | C16H10ClN3 |
| Molar mass | 279.73 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Synthesis
editReaction of the acyl hydrazide (1) and the carboximidate (2) gives compound (3). Heating this in the presence of sodium hydride cyclises this intermediate, forming the 1,2,4-triazole ring system of the drug. Oxidation of (4) with N-bromoacetamide yields lotrifen.[3][4]
References
edit- ^ a b Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 743–. ISBN 978-1-4757-2085-3.
- ^ Negwer M, Scharnow HG (4 October 2001). Organic-chemical drugs and their synonyms: (an international survey). Wiley-VCH. p. 1184. ISBN 978-3-527-30247-5.
- ^ "Lotrifen". chemdrug.com. Retrieved 2024-07-03.
- ^ US patent 4075341, Amedeo Omodei-Sale, Pietro Consonni and Leonard Lerner, "2-Substituted phenyl-5-triazols [5,1-a]isoquinoline compounds", issued 1978-02-21, assigned to Gruppo Lepetit S.P.A.
Further reading
edit- Lerner LJ (1989). "Development of novel embryotoxic compounds for interceptive fertility control in the dog". Journal of Reproduction and Fertility. Supplement. 39: 251–65. PMID 2621727.
- Wanke R, Hermanns W (January 1989). "Adverse effects of the antifertility agent DL 717-IT (Canocenta, Byk Gulden, FRG). Pathological findings in the rabbit". Research Communications in Chemical Pathology and Pharmacology. 63 (1): 69–80. PMID 2916081.