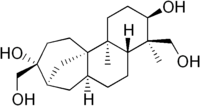
Aphidicolin is a tetracyclic diterpene antibiotic isolated from the fungus Cephalosporum aphidicola with antiviral and antimitotic properties. Aphidicolin is a reversible inhibitor of eukaryotic nuclear DNA replication. It blocks the cell cycle at early S phase. It is a specific inhibitor of DNA polymerase Alpha and Delta in eukaryotic cells and in some viruses (vaccinia[1][2] and herpesviruses) and an apoptosis inducer in HeLa cells. Natural aphidicolin is a secondary metabolite of the fungus Nigrospora oryzae.[3]
| |
| Names | |
|---|---|
| IUPAC name
(3R,4R,4aR,6aS,8R,9R,11aS,11bS)-4,9-bis(hydroxymethyl)-4,11b-dimethyltetradecahydro-8,11a-methanocyclohepta[a]naphthalene-3,9-diol
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.109.656 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C20H34O4 | |
| Molar mass | 338.48 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Bibliography
edit- Dhillon VS, Husain SA, Ray GN (2003). "Expression of aphidicolin-induced fragile sites and their relationship between genetic susceptibility in breast cancer, ovarian cancer, and non-small-cell lung cancer patients". Teratogenesis, Carcinogenesis, and Mutagenesis. Suppl 1: 35–45. doi:10.1002/tcm.10068. PMID 12616595.
- Mahy, Brian W J (2001). A dictionary of virology (3. ed.). San Diego, Calif. [u.a.]: Academic Press. p. 19. ISBN 978-0-12-465327-6.
References
edit- ^ DeFilippes, FM (Nov 1984). "Effect of aphidicolin on vaccinia virus: isolation of an aphidicolin-resistant mutant". Journal of Virology. 52 (2): 474–82. doi:10.1128/JVI.52.2.474-482.1984. PMC 254548. PMID 6436508.
- ^ Bucknall, R. A.; Moores, H.; Simms, R.; Hesp, B. (1 September 1973). "Antiviral Effects of Aphidicolin, a New Antibiotic Produced by Cephalosporium aphidicola". Antimicrobial Agents and Chemotherapy. 4 (3): 294–298. doi:10.1128/AAC.4.3.294. PMC 444544. PMID 4357181.
- ^ Aphidicolin product page Archived June 26, 2006, at the Wayback Machine from Fermentek