Benazepril, sold under the brand name Lotensin among others, is a medication used to treat high blood pressure, heart failure, and diabetic kidney disease.[2] It is a reasonable initial treatment for high blood pressure.[2] It is taken by mouth.[2] Versions are available as the combinations benazepril/hydrochlorothiazide and benazepril/amlodipine.[2]
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /bəˈnæzəprɪl/ |
| Trade names | Lotensin, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a692011 |
| License data |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 96.7% |
| Metabolism | Liver glucuronidation |
| Elimination half-life | 10-11 hours |
| Excretion | Kidney and bile duct |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
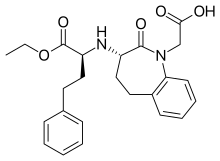
| Formula | C24H28N2O5 |
| Molar mass | 424.497 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Common side effects include feeling tired, dizziness, cough, and light-headedness with standing.[2] Serious side effects may include kidney problems, low blood pressure, high blood potassium, and angioedema.[2] Use in pregnancy may harm the baby, while use when breastfeeding may be safe.[3] It is an ACE inhibitor and works by decreasing renin-angiotensin-aldosterone system activity.[2]
Benazepril was patented in 1981 and came into medical use in 1990. It is available as a generic medication.[2] In 2021, it was the 147th most commonly prescribed medication in the United States, with more than 3 million prescriptions.[4][5]
Medical uses edit
Lotensin is indicated for the treatment of hypertension, to lower blood pressure.[1][2]
Side effects edit
The most common side effects patients experience are a headache or a chronic cough. The chronic cough develops in about 20% of people treated.[6]
Contraindications edit
Benazepril can harm the fetus.[7]
Dosage forms edit
It is also available in combination with hydrochlorothiazide, under the brand name Lotensin HCT, and with amlodipine (Lotrel).
Veterinary uses edit
Under the brand names Fortekor (Novartis) and VetACE (Jurox Animal Health),[citation needed] benazepril is used to treat congestive heart failure in dogs[8][9] and chronic kidney failure in cats and dogs.[10]
References edit
- ^ a b "Lotensin- benazepril hydrochloride tablet". DailyMed. 21 January 2019. Retrieved 11 February 2024.
- ^ a b c d e f g h i "Benazepril Hydrochloride Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 3 March 2019.
- ^ "Benazepril Pregnancy and Breastfeeding Warnings". Drugs.com. Retrieved 3 March 2019.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Benazepril - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- ^ Dykewicz MS (April 2004). "Cough and Angioedema From Angiotensin-Converting Enzyme Inhibitors: New Insights Into Mechanisms and Management". Medscape. Retrieved 2 April 2014.
- ^ "Lotensin package insert" (PDF). U.S. Food and Drug Administration (FDA). 2011. Retrieved 9 December 2020.
- ^ King JN, Mauron C, Kaiser G (December 1995). "Pharmacokinetics of the active metabolite of benazepril, benazeprilat, and inhibition of plasma angiotensin-converting enzyme activity after single and repeated administrations to dogs". American Journal of Veterinary Research. 56 (12): 1620–1628. PMID 8599524.
- ^ O'Grady MR, O'Sullivan ML, Minors SL, Horne R (2009). "Efficacy of benazepril hydrochloride to delay the progression of occult dilated cardiomyopathy in Doberman Pinschers". Journal of Veterinary Internal Medicine. 23 (5): 977–983. doi:10.1111/j.1939-1676.2009.0346.x. PMID 19572914.
- ^ "Fortekor Flavor Tabs (5 mg) (Canada) for Animal Use". Drugs.com. Retrieved 9 December 2020.